Toward safe lithium metal anode in rechargeable batteries: a review. Suspension electrolyte with modified Li + solvation environment for lithium metal batteries. Understanding and applying Coulombic efficiency in lithium metal batteries. Capturing the swelling of solid-electrolyte interphase in lithium metal batteries. Tailoring electrolyte solvation for Li metal batteries cycled at ultra-low temperature. Self-smoothing anode for achieving high-energy lithium metal batteries under realistic conditions. Our work provides insights into the interplay between the molecular structure of Li salts, their physicochemical properties and electrochemical performances. Under fast-cycling conditions (charging: 1.46 mA cm − 2, discharging: 3.66 mA cm − 2), pouch cells maintained 81% capacity after 100 cycles. Pouch cells of 310 Wh kg −1 achieved ~410 W kg −1 power density at the discharging current density of 6.59 mA cm −2. 
LiFEA-based carbonate electrolytes notably improved fast-cycling performances of Li | |NCM811 cells. It enables carbonate electrolytes with a large apparent donor number and Li + transference number and drives a self-cleaning mechanism for solid–electrolyte interphases, enhancing compatibility with Li-metal anodes even at high current densities. Herein we design an asymmetric Li salt, lithium 1,1,1-trifluoro- N-ethyl] methanesulfonamide (LiFEA) that possesses a pseudo-crown ether-like, folded molecular geometry. If your salt bridge is solid, like a lemon or a potato, just stick in the cathode and anode a small distance apart! When using a liquid electrolyte, hold your electrodes so that they both contact the liquid without touching each other.Conventional carbonate-based electrolytes with high corrosion towards Li metal result in massive dendrite growth and limited cycling life, particularly true for practical Li-metal batteries with high cathode loading (>3.5 mAh cm − 2). Experiment with produce items and non-toxic household liquids to see what works as a salt bridge. This is an easy experiment to play with at home! A penny and a piece of a soda can make good electrodes (buff the inside of the can with steel wool to remove the plastic coating and expose the aluminum). Ions flow through the Coke, which acts as a salt bridge. Electrons flow through the wires & multimeter. Gatorade, Coke, lemon juice, and salt water all do the trick, although commercial batteries more commonly rely on alkaline pastes.Ī tabletop battery with an aluminum anode and copper cathode. Substances like these are known as electrolytes. This path is called a salt bridge, and it can be made of anything that contains ions and conducts electricity when in liquid form. Ions on both sides of the reaction need a controlled escape path if a steady flow of electricity is to be maintained. If we mix all the anions and cations together, their charges equalize, and electrons no longer flow. If the copper anions build up at the cathode, their negative charge repels incoming electrons, and the redox reaction dies out. In our tabletop battery, aluminum donates electrons to copper, creating positively charged aluminum ions ( cations ) and negatively charged copper ions ( anions ). Electrons move through a wire circuit while ions flow through an electrolyte solution between the anode and the cathode. Completing a circuit around a battery gives electrons a path to follow and allows us to harvest power from their movement. 
Just like we can’t capture the power of the wind without putting a windmill in the way, we need to intercept the flow of electrons in a battery in order to get energy out. Strategic separation of the anode and cathode sets batteries apart from other redox reactions. We call the donor the anode and the recipient the cathode. In a redox reaction, one material gives electrons to another. 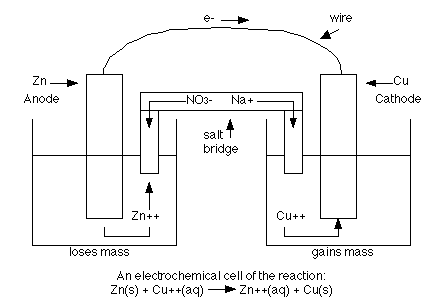
How do we get electrons going? Chemistry! Most batteries use a simple oxidation-reduction (or redox ) reaction to get electric charges moving. Batteries harness energy from the motion of electrons. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
